CogiLIMS®’s modular design allows you to activate only the modules that you wish to use, on demand. Begin your LIMS experience with the Analysis module and activate the other modules according to your specific needs and financial capacity.
GENERAL
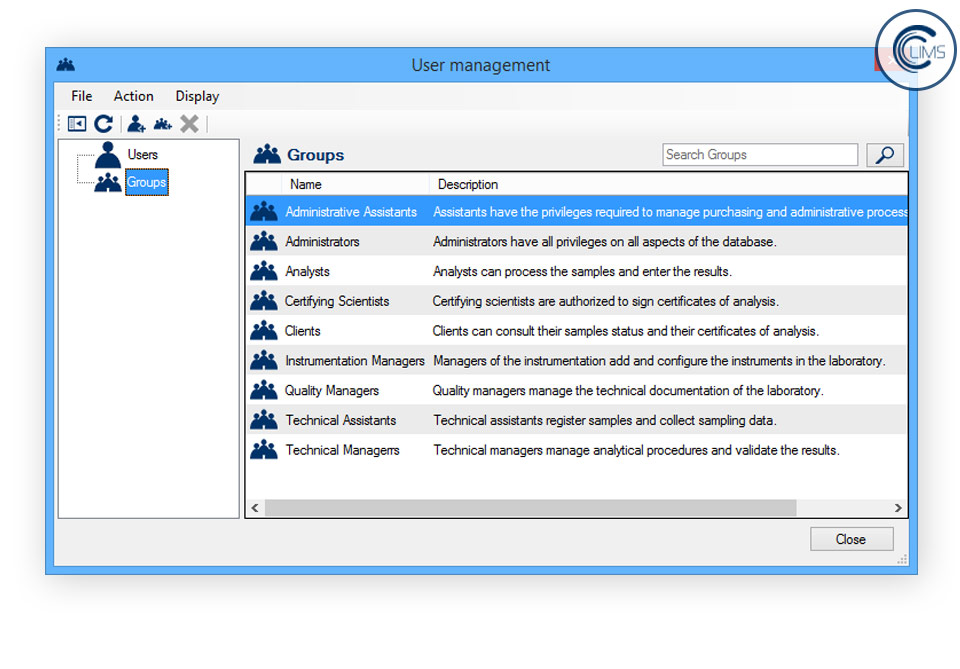
- Security management: Globally customize the security parameters of the client software according to your needs.
- User privilege management: Define the users’ access privileges by affiliating them to groups and adjust the groups’ privileges with an user-friendly client interface.
- Data import: The import of several reference lists upon their publication by regulation agencies allows CogiLIMS® to evolve and meet regulations continuously.
- Full-text search: Rapidly isolate the desired data using simple searches in all information panels.
- Export to spreadsheet: Isolate data with a search and export the search results to Microsoft Office Excel® for more flexible processing.
- Multilingual software (French/English): Generate documents according to your customers’ language preferences.
Analysis
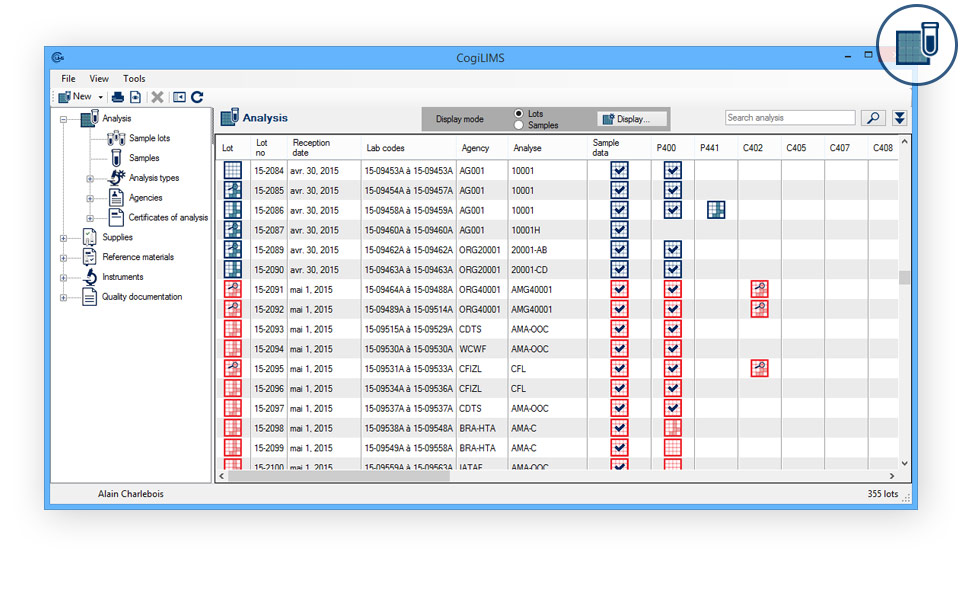
- Automated transfer of sample collection data (paperless DCFs): Collection documents received directly into the LIMS. Simplify your document management.
- Customer data: Contact information, document handling preferences and billing data.
- Flexible analysis procedures: Set or modify the analysis procedures directly into the client interface.
- Customizable types of analysis: Adapt the analyses to your customers’ specificities.
- Batch analysis programming: Assemble the samples in logical batches for more efficient processing.
- Generation of certificates of analysis in compliance with the World Anti-Doping Association and ISO 17025:2005 requirements.
- Result entry and audit trail with an intuitive graphical interface.
- Analysis sequences generation: Several types of analytical instruments are supported.
- Electronic transmission of results to the World Anti-Doping Association using XML, including all sample and analysis types and all analysis results.
- Billing of the analyses
Supplies
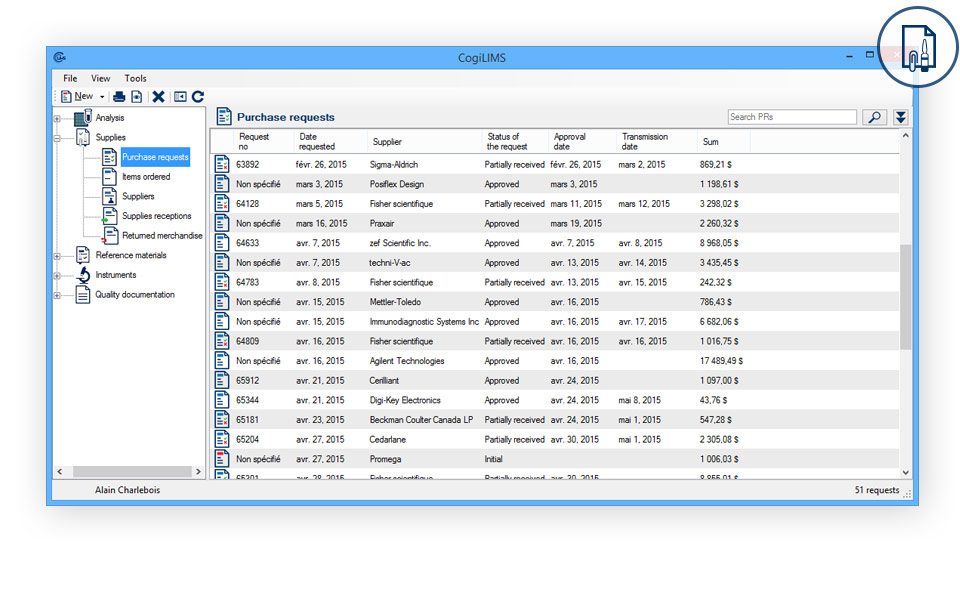
- Suppliers and list of approved supplies: Evaluate your supplies and block any unapproved purchase of supplies.
- Management of purchase requisitions: Register and approve purchase requisitions.
- Registration of received and returned supplies: Register the goods receipt and verify completeness of purchase requisitions.
INSTRUMENTS
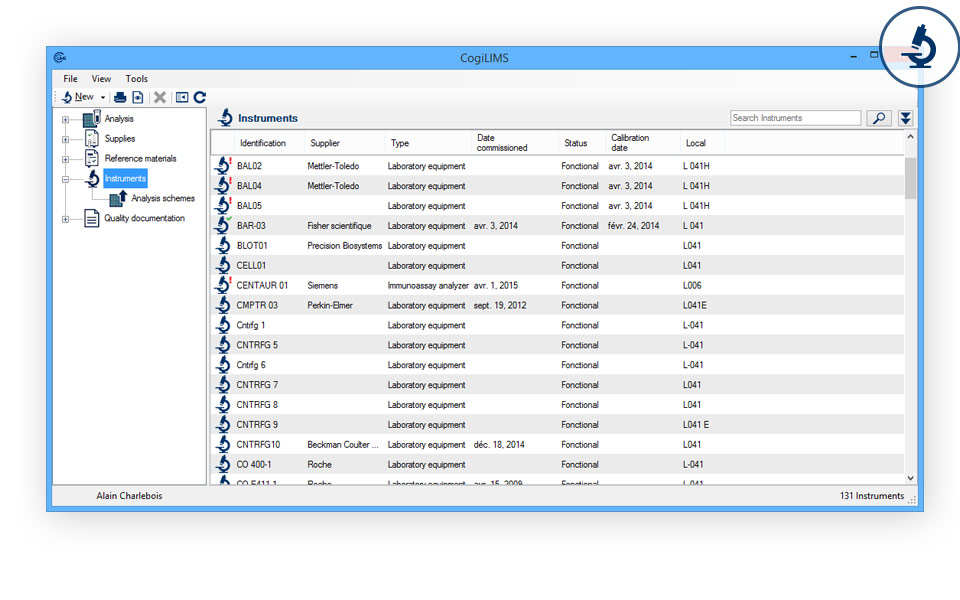
- Instrument management: Register the entry into service and the condition of your measuring instruments.
- Calibration status and dates: Keep an eye on the validity periods of the calibration certificates of your measuring instruments.
- Analysis schemes: Set useful analysis schemes for the generation of instrumental analysis sequences.
REFERENCE MATERIALS
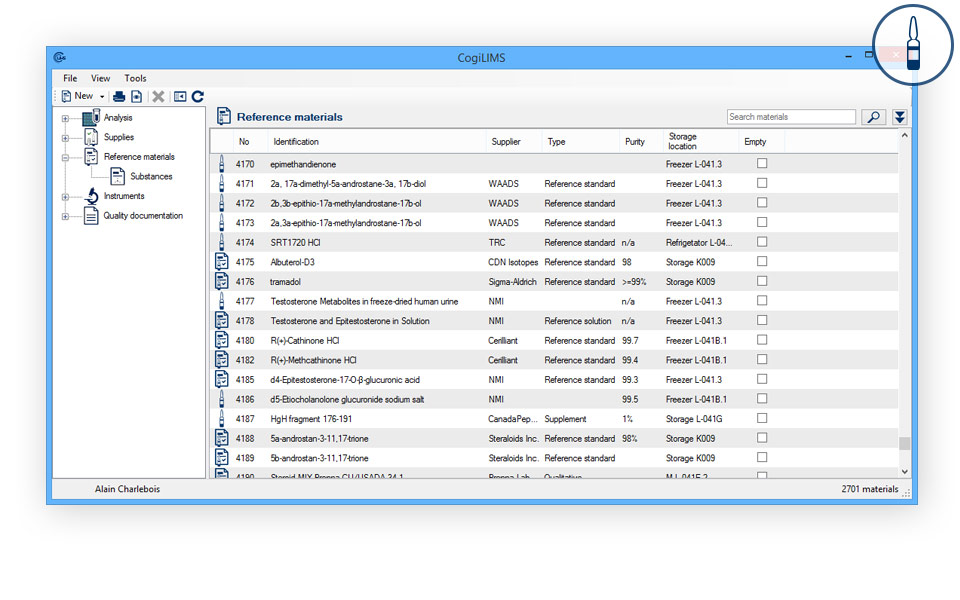
- Reference material management: Identify available reference material according to their physicochemical properties.
- Identification of controlled substances: Easily produce audit lists of all restricted use substances.
- Registration of initial quantity and of samplings:
- Inventory lists.
QUALITY MANAGEMENT
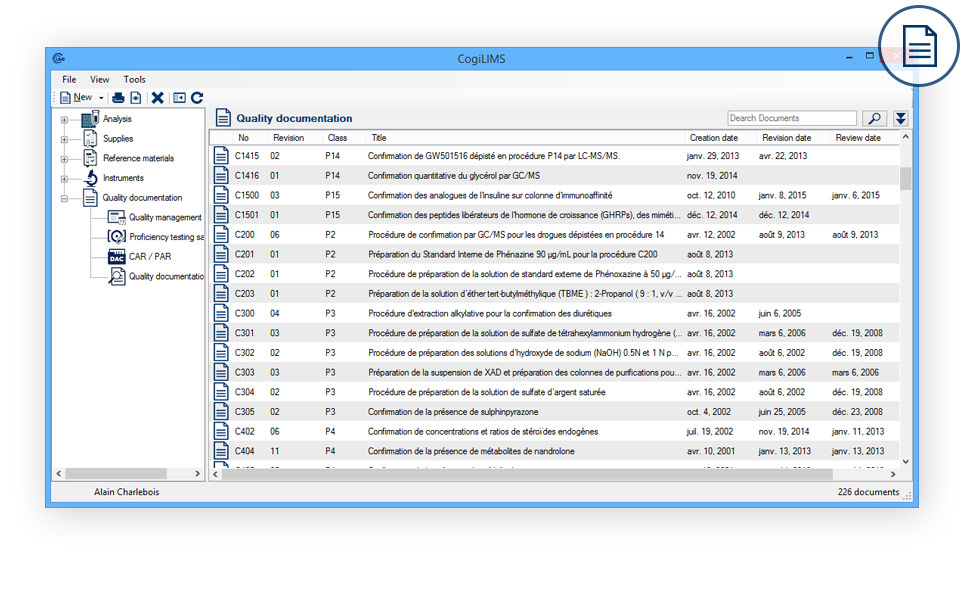
- Internal audits: Demonstrate internal audit coverage.
- Proficiency testing and interlaboratory comparisons: Document the proficiency tests’ results.
- Corrective or preventive action requests: Complete monitoring of the requests for corrective or preventive action.
- Document review management: Demonstrate your quality documentation management.
- Customer claims: Complete monitoring of customer complaints.

